Exercise-mediated reinnervation of skeletal muscle in elderly people: An update
HTML: 190
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Authors
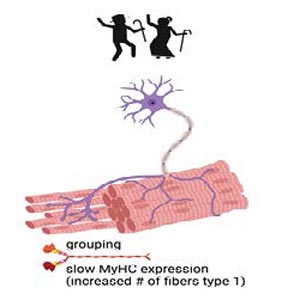
Sarcopenia is defined by the loss of muscle mass and function. In aging sarcopenia is due to mild chronic inflammation but also to fiber-intrinsic defects, such as mitochondrial dysfunction. Age-related sarcopenia is associated with physical disability and lowered quality of life. In addition to skeletal muscle, the nervous tissue is also affected in elderly people. With aging, type 2 fast fibers preferentially undergo denervation and are reinnervated by slow-twitch motor neurons. They spread forming new neuro-muscular junctions with the denervated fibers: the result is an increased proportion of slow fibers that group together since they are associated in the same motor unit. Grouping and fiber type shifting are indeed major histological features of aging skeletal muscle. Exercise has been proposed as an intervention for age-related sarcopenia due to its numerous beneficial effects on muscle mechanical and biochemical features. In 2013, a precursor study in humans was published in the European Journal of Translation Myology (formerly known as Basic and Applied Myology), highlighting the occurrence of reinnervation in the musculature of aged, exercise-trained individuals as compared to the matching control. This paper, entitled «Reinnervation of Vastus lateralis is increased significantly in seniors (70-years old) with a lifelong history of high-level exercise», is now being reprinted for the second issue of the «Ejtm Seminal Paper Series». In this short review we discuss those results in the light of the most recent advances confirming the occurrence of exercise-mediated reinnervation, ultimately preserving muscle structure and function in elderly people who exercise.
How to Cite
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.
Similar Articles
- Sabrina Conejeros-Lillo, Francisco Aguirre, Daniel Cabrera, Felipe Simon, Luis Peñailillo, Claudio Cabello-Verrugio, Role of the ubiquitin-proteasome system in the sarcopenic-like phenotype induced by CCL5/RANTES , European Journal of Translational Myology: Vol. 34 No. 1 (2024)
- Alessandra Renzini, Anna Benedetti, Marina Bouché, Leopoldo Silvestroni, Sergio Adamo, Viviana Moresi, Culture conditions influence satellite cell activation and survival of single myofibers , European Journal of Translational Myology: Vol. 28 No. 2 (2018)
- Ugo Carraro, EJTM3 is also covering Mobility and Medicine at large, an update , European Journal of Translational Myology: Vol. 28 No. 3 (2018)
- Ugo Carraro, From the Padua Muscle Days, the Basic and Applied Myology and the European Journal of Translational Myology to the A&CM Carraro Foundation for Translational Myology , European Journal of Translational Myology: Vol 27, No 3 (2017)
- Ugo Carraro, 2017Spring PaduaMuscleDays, roots and byproducts , European Journal of Translational Myology: Vol. 27 No. 2 (2017)
- Sergio Adamo, From Ejtm (European Journal of Translational Myology) to Ejt3M (European Journal of Translational Myology, Mobility, Medicine) , European Journal of Translational Myology: Vol. 28 No. 1 (2018)
You may also start an advanced similarity search for this article.

 https://doi.org/10.4081/ejtm.2022.10416
https://doi.org/10.4081/ejtm.2022.10416



