 https://doi.org/10.4081/ejtm.2020.9311
https://doi.org/10.4081/ejtm.2020.9311
Muscle hypertrophy and muscle strength: dependent or independent variables? A provocative review
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 9 September 2020
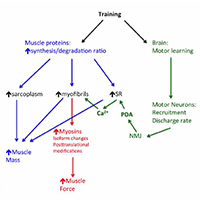
The question whether the muscle hypertrophy induced by resistance training, hormone administration or genetic manipulation is accompanied by a proportional increase in muscle strength is still open. This review summarizes and analyses data obtained in human and rodent muscles in studies that have monitored in parallel changes in muscle size and changes in muscle force, measured in isometric contractions in vivo, in isolated muscles ex vivo (in rodents) and in single muscle fibers. Although a general positive relation exists among the two variables, a number of studies show a clear dissociation with increase of muscle size with no change or even decrease in strength and, vice versa, increase in strength without increase in size. The possible mechanisms of such dissociation, which involves neural motor control and/or cellular and molecular adaptations of muscle fibers, are briefly discussed.
Downloads
Rasch PJ. The problem of muscle hypertrophy: a review. J Am Osteopath Assoc 1955;54:525–8.
Moritani T, deVries HA. Neural factors versus hypertrophy in the time course of muscle strength gain. Am J Phys Med 1979;58:115–30.
Buckner SL, Dankel SJ, Mattocks KT, et al. The problem Of muscle hypertrophy: Revisited. Muscle Nerve 2016;54:1012–4. https://doi.org/10.1002/mus.25420. DOI: https://doi.org/10.1002/mus.25420
Loenneke JP, Buckner SL, Dankel SJ, et al. Exercise-Induced Changes in Muscle Size do not Contribute to Exercise-Induced Changes in Muscle Strength. Sports Med 2019;49:987–91. https://doi.org/10.1007/s40279-019-01106-9. DOI: https://doi.org/10.1007/s40279-019-01106-9
Loenneke JP, Dankel SJ, Bell ZW, et al. Is muscle growth a mechanism for increasing strength? Med Hypotheses 2019;125:51–6. https://doi.org/10.1016/j.mehy.2019.02.030. DOI: https://doi.org/10.1016/j.mehy.2019.02.030
Roberts MD, Haun CT, Vann CG, et al. Sarcoplasmic Hypertrophy in Skeletal Muscle: A Scientific ‘Unicorn’ or Resistance Training Adaptation? Front Physiol 2020;11:816. https://doi.org/10.3389/fphys.2020.00816. DOI: https://doi.org/10.3389/fphys.2020.00816
Tesch PA. Training for bodybuilding. In: Komi PV, editor. Strength and power in sport. 2nd ed. London: Blackwell Scientific,1992; n.d. p. 370–80.
Fry AC. The role of resistance exercise intensity on muscle fibre adaptations. Sports Med 2004;34:663–79. https://doi.org/10.2165/00007256-200434100-00004. DOI: https://doi.org/10.2165/00007256-200434100-00004
Ikegawa S, Funato K, Tsunoda N, et al. Muscle force per cross-sectional area is inversely related with pennation angle in strength trained athletes. J Strength Cond Res 2008;22:128–31. https://doi.org/10.1519/JSC.0b013e31815f2fd3. DOI: https://doi.org/10.1519/JSC.0b013e31815f2fd3
Ahtiainen JP, Walker S, Peltonen H, et al. Heterogeneity in resistance training-induced muscle strength and mass responses in men and women of different ages. Age (Dordr) 2016;38:10. https://doi.org/10.1007/s11357-015-9870-1. DOI: https://doi.org/10.1007/s11357-015-9870-1
Erskine RM, Fletcher G, Folland JP. The contribution of muscle hypertrophy to strength changes following resistance training. Eur J Appl Physiol 2014;114:1239–49. https://doi.org/10.1007/s00421-014-2855-4. DOI: https://doi.org/10.1007/s00421-014-2855-4
Maughan RJ, Watson JS, Weir J. Strength and cross-sectional area of human skeletal muscle. J Physiol (Lond) 1983;338:37–49. https://doi.org/10.1113/jphysiol.1983.sp014658. DOI: https://doi.org/10.1113/jphysiol.1983.sp014658
Alway SE, Stray-Gundersen J, Grumbt WH, et al. Muscle cross-sectional area and torque in resistance-trained subjects. Eur J Appl Physiol Occup Physiol 1990;60:86–90. https://doi.org/10.1007/BF00846026. DOI: https://doi.org/10.1007/BF00846026
Schoenfeld BJ, Contreras B, Krieger J, et al. Resistance Training Volume Enhances Muscle Hypertrophy but Not Strength in Trained Men. Med Sci Sports Exerc 2019;51:94–103. https://doi.org/10.1249/MSS.0000000000001764. DOI: https://doi.org/10.1249/MSS.0000000000001764
Mason J, Frazer AK, Pearce AJ, et al. Determining the early corticospinal-motoneuronal responses to strength training: a systematic review and meta-analysis. Rev Neurosci 2019;30:463–76. https://doi.org/10.1515/revneuro-2018-0054. DOI: https://doi.org/10.1515/revneuro-2018-0054
Seynnes OR, Kamandulis S, Kairaitis R, et al. Effect of androgenic-anabolic steroids and heavy strength training on patellar tendon morphological and mechanical properties. J Appl Physiol 2013;115:84–9. https://doi.org/10.1152/japplphysiol.01417.2012. DOI: https://doi.org/10.1152/japplphysiol.01417.2012
Widrick JJ, Stelzer JE, Shoepe TC, et al. Functional properties of human muscle fibers after short-term resistance exercise training. Am J Physiol Regul Integr Comp Physiol 2002;283:R408-416. https://doi.org/10.1152/ajpregu.00120.2002. DOI: https://doi.org/10.1152/ajpregu.00120.2002
Pansarasa O, Rinaldi C, Parente V, et al. Resistance training of long duration modulates force and unloaded shortening velocity of single muscle fibres of young women. J Electromyogr Kinesiol 2009;19:e290-300. https://doi.org/10.1016/j.jelekin.2008.07.007. DOI: https://doi.org/10.1016/j.jelekin.2008.07.007
Shoepe TC, Stelzer JE, Garner DP, et al. Functional adaptability of muscle fibers to long-term resistance exercise. Med Sci Sports Exerc 2003;35:944–51. https://doi.org/10.1249/01.MSS.0000069756.17841.9E. DOI: https://doi.org/10.1249/01.MSS.0000069756.17841.9E
D’Antona G, Lanfranconi F, Pellegrino MA, et al. Skeletal muscle hypertrophy and structure and function of skeletal muscle fibres in male body builders. J Physiol (Lond) 2006;570:611–27. https://doi.org/10.1113/jphysiol.2005.101642. DOI: https://doi.org/10.1113/jphysiol.2005.101642
Meijer JP, Jaspers RT, Rittweger J, et al. Single muscle fibre contractile properties differ between body-builders, power athletes and control subjects. Exp Physiol 2015;100:1331–41. https://doi.org/10.1113/EP085267. DOI: https://doi.org/10.1113/EP085267
Claassen H, Gerber C, Hoppeler H, et al. Muscle filament spacing and short-term heavy-resistance exercise in humans. J Physiol (Lond) 1989;409:491–5. https://doi.org/10.1113/jphysiol.1989.sp017509. DOI: https://doi.org/10.1113/jphysiol.1989.sp017509
MacDougall JD, Sale DG, Elder GCB, et al. Muscle ultrastructural characteristics of elite powerlifters and bodybuilders. Europ J Appl Physiol 1982;48:117–26. https://doi.org/10.1007/BF00421171. DOI: https://doi.org/10.1007/BF00421171
Toth MJ, Miller MS, VanBuren P, et al. Resistance training alters skeletal muscle structure and function in human heart failure: effects at the tissue, cellular and molecular levels. J Physiol (Lond) 2012;590:1243–59. https://doi.org/10.1113/jphysiol.2011.219659. DOI: https://doi.org/10.1113/jphysiol.2011.219659
MacDougall JD, Ward GR, Sale DG, et al. Biochemical adaptation of human skeletal muscle to heavy resistance training and immobilization. J Appl Physiol Respir Environ Exerc Physiol 1977;43:700–3. https://doi.org/10.1152/jappl.1977.43.4.700. DOI: https://doi.org/10.1152/jappl.1977.43.4.700
Haun CT, Vann CG, Osburn SC, et al. Muscle fiber hypertrophy in response to 6 weeks of high-volume resistance training in trained young men is largely attributed to sarcoplasmic hypertrophy. PLoS ONE 2019;14:e0215267. https://doi.org/10.1371/journal.pone.0215267. DOI: https://doi.org/10.1371/journal.pone.0215267
Brook MS, Wilkinson DJ, Mitchell WK, et al. Skeletal muscle hypertrophy adaptations predominate in the early stages of resistance exercise training, matching deuterium oxide-derived measures of muscle protein synthesis and mechanistic target of rapamycin complex 1 signaling. The FASEB Journal 2015;29:4485–96. https://doi.org/10.1096/fj.15-273755. DOI: https://doi.org/10.1096/fj.15-273755
Ato S, Tsushima D, Isono Y, et al. The Effect of Changing the Contraction Mode During Resistance Training on mTORC1 Signaling and Muscle Protein Synthesis. Front Physiol 2019;10:406. https://doi.org/10.3389/fphys.2019.00406. DOI: https://doi.org/10.3389/fphys.2019.00406
Wilkinson DJ, Franchi MV, Brook MS, et al. A validation of the application of D(2)O stable isotope tracer techniques for monitoring day-to-day changes in muscle protein subfraction synthesis in humans. Am J Physiol Endocrinol Metab 2014;306:E571-579. https://doi.org/10.1152/ajpendo.00650.2013. DOI: https://doi.org/10.1152/ajpendo.00650.2013
Vissing K, Groennebaek T, Wernbom M, et al. Myocellular Adaptations to Low-Load Blood Flow Restricted Resistance Training. Exercise and Sport Sciences Reviews 2020;Publish Ahead of Print: https://doi.org/10.1249/JES.0000000000000231. DOI: https://doi.org/10.1249/JES.0000000000000231
Nielsen JL, Aagaard P, Bech RD, et al. Proliferation of myogenic stem cells in human skeletal muscle in response to low-load resistance training with blood flow restriction. J Physiol 2012;590:4351–61. https://doi.org/10.1113/jphysiol.2012.237008. DOI: https://doi.org/10.1113/jphysiol.2012.237008
Fitts RH, Peters JR, Dillon EL, et al. Weekly versus monthly testosterone administration on fast and slow skeletal muscle fibers in older adult males. J Clin Endocrinol Metab 2015;100:E223-231. https://doi.org/10.1210/jc.2014-2759. DOI: https://doi.org/10.1210/jc.2014-2759
Lamboley CR, Xu H, Dutka TL, et al. Effect of androgen deprivation therapy on the contractile properties of type I and type II skeletal muscle fibres in men with non-metastatic prostate cancer. Clin Exp Pharmacol Physiol 2018;45:146–54. https://doi.org/10.1111/1440-1681.12873. DOI: https://doi.org/10.1111/1440-1681.12873
Roy RR, Wilson R, Edgerton VR. Architectural and mechanical properties of the rat adductor longus: response to weight-lifting training. Anat Rec 1997;247:170–8. https://doi.org/10.1002/(SICI)1097-0185(199702)247:2<170::AID-AR3>3.0.CO;2-1. DOI: https://doi.org/10.1002/(SICI)1097-0185(199702)247:2<170::AID-AR3>3.0.CO;2-1
Goldberg AL. Work-induced growth of skeletal muscle in normal and hypophysectomized rats. Am J Physiol 1967;213:1193–8. https://doi.org/10.1152/ajplegacy.1967.213.5.1193. DOI: https://doi.org/10.1152/ajplegacy.1967.213.5.1193
Hubbard RW, Ianuzzo CD, Mathew WT, et al. Compensatory adaptations of skeletal muscle composition to a long-term functional overload. Growth 1975;39:85–93.
Kandarian SC, White TP. Mechanical deficit persists during long-term muscle hypertrophy. J Appl Physiol 1990;69:861–7. https://doi.org/10.1152/jappl.1990.69.3.861. DOI: https://doi.org/10.1152/jappl.1990.69.3.861
Pérez-Schindler J, Summermatter S, Santos G, et al. The transcriptional coactivator PGC-1α is dispensable for chronic overload-induced skeletal muscle hypertrophy and metabolic remodeling. Proc Natl Acad Sci USA 2013;110:20314–9. https://doi.org/10.1073/pnas.1312039110. DOI: https://doi.org/10.1073/pnas.1312039110
Mendias CL, Schwartz AJ, Grekin JA, et al. Changes in muscle fiber contractility and extracellular matrix production during skeletal muscle hypertrophy. J Appl Physiol 2017;122:571–9. https://doi.org/10.1152/japplphysiol.00719.2016. DOI: https://doi.org/10.1152/japplphysiol.00719.2016
Antonio-Santos J, Ferreira DJS, Gomes Costa GL, et al. Resistance Training Alters the Proportion of Skeletal Muscle Fibers but Not Brain Neurotrophic Factors in Young Adult Rats. J Strength Cond Res 2016;30:3531–8. https://doi.org/10.1519/JSC.0000000000001449. DOI: https://doi.org/10.1519/JSC.0000000000001449
Duncan ND, Williams DA, Lynch GS. Adaptations in rat skeletal muscle following long-term resistance exercise training. Eur J Appl Physiol Occup Physiol 1998;77:372–8. https://doi.org/10.1007/s004210050347. DOI: https://doi.org/10.1007/s004210050347
Cui D, Drake JC, Wilson RJ, et al. A novel voluntary weightlifting model in mice promotes muscle adaptation and insulin sensitivity with simultaneous enhancement of autophagy and mTOR pathway. FASEB J 2020;34:7330–44. https://doi.org/10.1096/fj.201903055R. DOI: https://doi.org/10.1096/fj.201903055R
Dungan CM, Murach KA, Frick KK, et al. Elevated myonuclear density during skeletal muscle hypertrophy in response to training is reversed during detraining. Am J Physiol, Cell Physiol 2019;316:C649–54. https://doi.org/10.1152/ajpcell.00050.2019. DOI: https://doi.org/10.1152/ajpcell.00050.2019
Seldeen KL, Lasky G, Leiker MM, et al. High Intensity Interval Training Improves Physical Performance and Frailty in Aged Mice. J Gerontol A Biol Sci Med Sci 2018;73:429–37. https://doi.org/10.1093/gerona/glx120. DOI: https://doi.org/10.1093/gerona/glx120
Goh Q, Song T, Petrany MJ, et al. Myonuclear accretion is a determinant of exercise-induced remodeling in skeletal muscle. Elife 2019;8:. https://doi.org/10.7554/eLife.44876. DOI: https://doi.org/10.7554/eLife.44876
Tobin C, Joubert Y. Testosterone-induced development of the rat levator ani muscle. Dev Biol 1991;146:131–8. https://doi.org/10.1016/0012-1606(91)90453-a. DOI: https://doi.org/10.1016/0012-1606(91)90453-A
Ye F, McCoy SC, Ross HH, et al. Transcriptional regulation of myotrophic actions by testosterone and trenbolone on androgen-responsive muscle. Steroids 2014;87:59–66. https://doi.org/10.1016/j.steroids.2014.05.024. DOI: https://doi.org/10.1016/j.steroids.2014.05.024
Ustünel I, Akkoyunlu G, Demir R. The effect of testosterone on gastrocnemius muscle fibres in growing and adult male and female rats: a histochemical, morphometric and ultrastructural study. Anat Histol Embryol 2003;32:70–9. https://doi.org/10.1046/j.1439-0264.2003.00441.x. DOI: https://doi.org/10.1046/j.1439-0264.2003.00441.x
Axell A-M, MacLean HE, Plant DR, et al. Continuous testosterone administration prevents skeletal muscle atrophy and enhances resistance to fatigue in orchidectomized male mice. Am J Physiol Endocrinol Metab 2006;291:E506-516. https://doi.org/10.1152/ajpendo.00058.2006. DOI: https://doi.org/10.1152/ajpendo.00058.2006
Zeman RJ, Ludemann R, Easton TG, et al. Slow to fast alterations in skeletal muscle fibers caused by clenbuterol, a beta 2-receptor agonist. Am J Physiol 1988;254:E726-732. https://doi.org/10.1152/ajpendo.1988.254.6.E726. DOI: https://doi.org/10.1152/ajpendo.1988.254.6.E726
Lynch GS, Hayes A, Campbell SP, et al. Effects of beta 2-agonist administration and exercise on contractile activation of skeletal muscle fibers. J Appl Physiol 1996;81:1610–8. https://doi.org/10.1152/jappl.1996.81.4.1610. DOI: https://doi.org/10.1152/jappl.1996.81.4.1610
Polla B, Cappelli V, Morello F, et al. Effects of the beta(2)-agonist clenbuterol on respiratory and limb muscles of weaning rats. Am J Physiol Regul Integr Comp Physiol 2001;280:R862-869. https://doi.org/10.1152/ajpregu.2001.280.3.R862. DOI: https://doi.org/10.1152/ajpregu.2001.280.3.R862
Kim J, Grotegut CA, Wisler JW, et al. β-arrestin 1 regulates β2-adrenergic receptor-mediated skeletal muscle hypertrophy and contractility. Skelet Muscle 2018;8:39. https://doi.org/10.1186/s13395-018-0184-8. DOI: https://doi.org/10.1186/s13395-018-0184-8
Kambadur R, Sharma M, Smith TP, et al. Mutations in myostatin (GDF8) in double-muscled Belgian Blue and Piedmontese cattle. Genome Res 1997;7:910–6. https://doi.org/10.1101/gr.7.9.910. DOI: https://doi.org/10.1101/gr.7.9.910
Amthor H, Macharia R, Navarrete R, et al. Lack of myostatin results in excessive muscle growth but impaired force generation. PNAS 2007;104:1835–40. https://doi.org/10.1073/pnas.0604893104. DOI: https://doi.org/10.1073/pnas.0604893104
Bodnár D, Geyer N, Ruzsnavszky O, et al. Hypermuscular mice with mutation in the myostatin gene display altered calcium signalling. J Physiol (Lond) 2014;592:1353–65. https://doi.org/10.1113/jphysiol.2013.261958. DOI: https://doi.org/10.1113/jphysiol.2013.261958
Blaauw B, Canato M, Agatea L, et al. Inducible activation of Akt increases skeletal muscle mass and force without satellite cell activation. FASEB J 2009;23:3896–905. https://doi.org/10.1096/fj.09-131870. DOI: https://doi.org/10.1096/fj.09-131870
Musarò A, McCullagh K, Paul A, et al. Localized Igf-1 transgene expression sustains hypertrophy and regeneration in senescent skeletal muscle. Nat Genet 2001;27:195–200. https://doi.org/10.1038/84839. DOI: https://doi.org/10.1038/84839
Ascenzi F, Barberi L, Dobrowolny G, et al. Effects of IGF-1 isoforms on muscle growth and sarcopenia. Aging Cell 2019;18:e12954. https://doi.org/10.1111/acel.12954. DOI: https://doi.org/10.1111/acel.12954
Gonzalez E, Messi ML, Zheng Z, et al. Insulin-like growth factor-1 prevents age-related decrease in specific force and intracellular Ca2+ in single intact muscle fibres from transgenic mice. J Physiol (Lond) 2003;552:833–44. https://doi.org/10.1113/jphysiol.2003.048165. DOI: https://doi.org/10.1113/jphysiol.2003.048165
Colombini B, Benelli G, Nocella M, et al. Mechanical properties of intact single fibres from wild-type and MLC/mIgf-1 transgenic mouse muscle. J Muscle Res Cell Motil 2009;30:199–207. https://doi.org/10.1007/s10974-009-9187-8. DOI: https://doi.org/10.1007/s10974-009-9187-8
Percario V, Boncompagni S, Protasi F, et al. Mechanical parameters of the molecular motor myosin II determined in permeabilised fibres from slow and fast skeletal muscles of the rabbit. J Physiol (Lond) 2018;596:1243–57. https://doi.org/10.1113/JP275404. DOI: https://doi.org/10.1113/JP275404
Schiaffino S, Reggiani C. Fiber types in mammalian skeletal muscles. Physiol Rev 2011;91:1447–531. https://doi.org/10.1152/physrev.00031.2010. DOI: https://doi.org/10.1152/physrev.00031.2010
Canepari M, Rossi R, Pellegrino MA, et al. Effects of resistance training on myosin function studied by the in vitro motility assay in young and older men. J Appl Physiol 2005;98:2390–5. https://doi.org/10.1152/japplphysiol.01103.2004. DOI: https://doi.org/10.1152/japplphysiol.01103.2004
Marcucci L, Washio T, Yanagida T. Titin-mediated thick filament activation, through a mechanosensing mechanism, introduces sarcomere-length dependencies in mathematical models of rat trabecula and whole ventricle. Scientific Reports 2017;7:5546. https://doi.org/10.1038/s41598-017-05999-2. DOI: https://doi.org/10.1038/s41598-017-05999-2
Irving M. Regulation of Contraction by the Thick Filaments in Skeletal Muscle. Biophys J 2017;113:2579–94. https://doi.org/10.1016/j.bpj.2017.09.037. DOI: https://doi.org/10.1016/j.bpj.2017.09.037
Lehman W. Thin Filament Structure and the Steric Blocking Model. Compr Physiol 2016;6:1043–69. https://doi.org/10.1002/cphy.c150030. DOI: https://doi.org/10.1002/cphy.c150030
Sirvent P, Douillard A, Galbes O, et al. Effects of chronic administration of clenbuterol on contractile properties and calcium homeostasis in rat extensor digitorum longus muscle. PLoS ONE 2014;9:e100281. https://doi.org/10.1371/journal.pone.0100281. DOI: https://doi.org/10.1371/journal.pone.0100281
Py G, Ramonatxo C, Sirvent P, et al. Chronic clenbuterol treatment compromises force production without directly altering skeletal muscle contractile machinery. J Physiol (Lond) 2015;593:2071–84. https://doi.org/10.1113/jphysiol.2014.287060. DOI: https://doi.org/10.1113/jphysiol.2014.287060
Folland JP, Williams AG. The adaptations to strength training : morphological and neurological contributions to increased strength. Sports Med 2007;37:145–68. https://doi.org/10.2165/00007256-200737020-00004. DOI: https://doi.org/10.2165/00007256-200737020-00004
Deschenes MR, Tufts HL, Oh J, et al. Effects of exercise training on neuromuscular junctions and their active zones in young and aged muscles. Neurobiol Aging 2020;95:1–8. https://doi.org/10.1016/j.neurobiolaging.2020.07.001. DOI: https://doi.org/10.1016/j.neurobiolaging.2020.07.001
Gollnick PD, Timson BF, Moore RL, et al. Muscular enlargement and number of fibers in skeletal muscles of rats. Journal of Applied Physiology 1981;50:936–43. https://doi.org/10.1152/jappl.1981.50.5.936. DOI: https://doi.org/10.1152/jappl.1981.50.5.936
Franchi MV, Atherton PJ, Reeves ND, et al. Architectural, functional and molecular responses to concentric and eccentric loading in human skeletal muscle. Acta Physiol (Oxf) 2014;210:642–54. https://doi.org/10.1111/apha.12225. DOI: https://doi.org/10.1111/apha.12225
Reeves ND, Narici MV, Maganaris CN. Myotendinous plasticity to ageing and resistance exercise in humans. Exp Physiol 2006;91:483–98. https://doi.org/10.1113/expphysiol.2005.032896. DOI: https://doi.org/10.1113/expphysiol.2005.032896
Reeves ND, Narici MV, Maganaris CN. In vivo human muscle structure and function: adaptations to resistance training in old age. Exp Physiol 2004;89:675–89. https://doi.org/10.1113/expphysiol.2004.027797. DOI: https://doi.org/10.1113/expphysiol.2004.027797
How to Cite
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

