 https://doi.org/10.4081/ejtm.2026.15055
https://doi.org/10.4081/ejtm.2026.15055
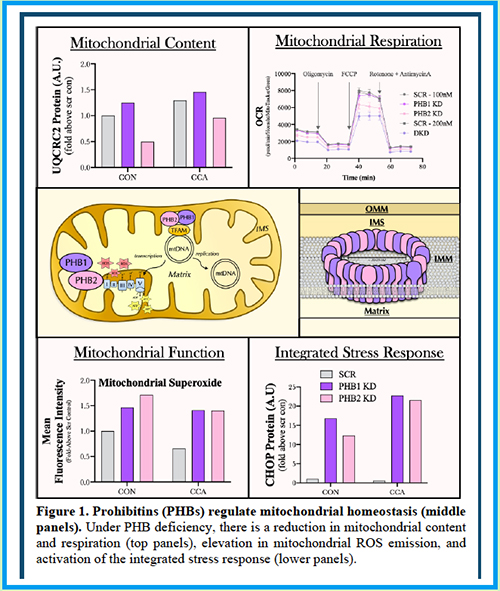
Abstract 056 | Role of prohibitin isoforms in mitochondrial function with aging and exercise
Sabrina Champsi, David A. Hood | Muscle Health Research Centre, School of Kinesiology and Health science, York University, Toronto, Canada.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Vainshtein A, Tryon LD, Pauly M, Hood DA. Role of PGC-1a during acute exercise-induced autophagy and mitophagy in skeletal muscle. Am J Physiol Cell Physiol. 2015 May 1;308(9): c710-9. Doi: 10.1152/ajpcell.00380.2014. Epub 2015 Feb 11. PMID: 25673772; PMCID: PMC4420796. DOI: https://doi.org/10.1152/ajpcell.00380.2014
2. Hyatt, H, Deminice R, Yoshihara T, Powers SK. Mitochondrial dysfunction induces muscle atrophy during prolonged inactivity: A review of the causes and effects. Arch Biochem Biophys. 2019 Feb 15;662:49-60. doi: DOI: https://doi.org/10.1016/j.abb.2018.11.005
10.1016/j.abb.2018.11.005. Epub 2018 Nov 16. PMID: 30452895; PMCID: PMC6783132.
3. Ljubicic V, Joseph AM, Adhihetty PJ, Huang JH, Saleem A, Uguccioni G, Hood DA. Molecular basis for an attenuated mitochondrial adaptive plasticity in aged skeletal muscle. Aging (Albany NY). 3009 Sep 12;1(9):818-30. Doi: 10.18632/aging.100083. PMID: 20157569; PMCID: PMC2815739. DOI: https://doi.org/10.18632/aging.100083
4. Carter HN, Pauly M, Tyron LD, Hood DA. Effect of contractile activity on PGC-1 transcription in young and aged skeletal muscle. J Appl Physiol (1985). 2018 Jun1;124(6):1605-1615. Doi: 10.1152/japplphysiol.01110.2017. Epub 2018 Mar 15. PMID: 29543139. DOI: https://doi.org/10.1152/japplphysiol.01110.2017
5. Chen CCW, Erlich AT, Hood DA. Role of Parkin and endurance training on mitochondrial turnover in skeletal muscle. Skelet Muscle. 2018 Mar 17;8(1): 10. Doi: 10.1186/s13395-018-0157-y. PMID: 29549884; PMCID: PMC5857114. DOI: https://doi.org/10.1186/s13395-018-0157-y
6. Ikonen E, Fiedler K, Parton RG, Simons K. Prohibitin, an antiproliferative protein, is localized to mitochondria. FEBS Lett. 1995 Jan 30;358(3): 273-7. doi: 10.1016/0014-5793(94)01444-6. PMID: 7843414. DOI: https://doi.org/10.1016/0014-5793(94)01444-6
7. Merkwirth C, Martinelli P, Korwitz A, Morbin M, Brönneke HS, Jordan SD, Rugarli El, Langer T. Loss of prohibitin membrane scaffolds impairs mitochondrial architecture and leads to tau hyperphosphorylation and
neurodegeneration. PLos Genet. 2012;8(11):e1003021. doi: 10.1371/journal.pgen.1003021. Epub 2012 Nov 8. PMID: 23144624; PMCID: PMC3493444.
8. Yang M, Audureyimu M, Wang X, Zhou Y, Zhang Y, Ren J. PHB2 ameliorates Doxorubicin-induced cardiomyopathy through interaction with NDUFV2 and restoration of mitochondrial complex I function. Redox Biol. 2023 Sep;65:102812. doi: 10.1016/j.redox.2023.102812. Epub 2012 Jul 12. PMID: 37451140; PMCID: PMC10366351. DOI: https://doi.org/10.1016/j.redox.2023.102812
9. Wei Y, Chiang WC, Sumpter R Jr, Mishra P, Levine B. Prohibitin 2 is an Inner Mitochondrial Membrane Mitophagy Receptor. Cell. 2017 Jan 12;168(1-2):224-238.e10. doi: 10.1016/j.cell.2016.11.042. Epub 2016 Dec 22. PMID: 28017329; PMCID: PMC5235968. DOI: https://doi.org/10.1016/j.cell.2016.11.042
10. Kasashima K, Sumitani M, Satoh M, Endo H. Human prohibitin 1 maintains the organization and stability of the mitochondrial nuceleoids. Exp Cell Res. 2008 Mar 10;314(5):988-96. Doi: 10.1016/j.yexcr.2008.01.005. Epub 2008 Jan 16. PMID: 18258228. DOI: https://doi.org/10.1016/j.yexcr.2008.01.005
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

