 https://doi.org/10.4081/ejtm.2026.15054
https://doi.org/10.4081/ejtm.2026.15054
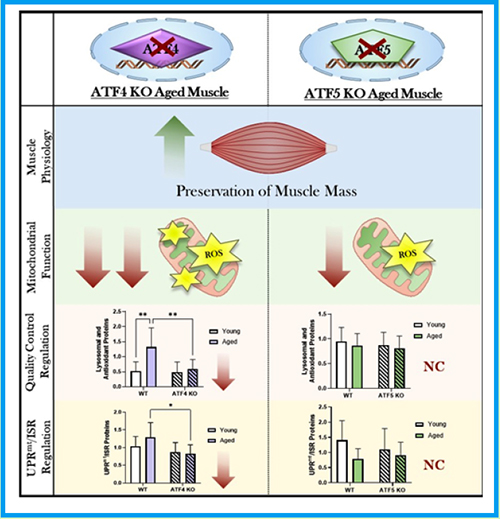
Abstract 055 | The importance of mitochondrial stress response regulators in aged muscle
Victoria C. Sanfrancesco, David A. Hood | Muscle Health Research Centre, School of Kinesiology and Health science, York University, Toronto, Canada.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Kotiadis VN, Duchen MR, Osellame LD. Mitochondrial quality control and communications with the nucleus are important in maintaining mitochondrial function and cell health. Biochim Biophys Acta. 2014 Apr;1840(4):1254-65. doi: 10.1016/j.bbagen.2013.10.041. Epub 2013 Nov 6. PMID: 24211250; PMCID: PMC3970188.
2. Anderson NS, Haynes CM. Folding the Mitochondrial UPR into the Integrated Stress Response. Trends Cell Biol. 2020 Jun;30(6):428-439. doi: 10.1016/j.tcb.2020.03.001. Epub 2020 Apr 2. PMID: 32413314; PMCID: PMC7230072.
3. Quirós PM, Prado MA, Zamboni N, D'Amico D, Williams RW, Finley D, Gygi SP, Auwerx J. Multi-omics analysis identifies ATF4 as a key regulator of the mitochondrial stress response in mammals. J Cell Biol. 2017 Jul 3;216(7):2027-2045. doi: 10.1083/jcb.201702058. Epub 2017 May 31. PMID: 28566324; PMCID: PMC5496626.
4. Slavin MB, Kumari R, Hood DA. ATF5 is a regulator of exercise-induced mitochondrial quality control in skeletal muscle. Mol Metab. 2022 Dec;66:101623. doi: 10.1016/j.molmet.2022.101623. Epub 2022 Nov 1. PMID: 36332794; PMCID: PMC9661517.
5. Memme JM, Sanfrancesco VC, Hood DA. Activating transcription factor 4 regulates mitochondrial content, morphology, and function in differentiating skeletal muscle myotubes. Am J Physiol Cell Physiol. 2023 Jul 1;325(1):C224-C242. doi: 10.1152/ajpcell.00080.2023. Epub 2023 Jun 5. PMID: 37273238.
6. Marzetti E, Calvani R, Coelho-Junior HJ, Landi F, Picca A. Defective mitochondrial quality control in the aging of skeletal muscle. Mech Ageing Dev. 2025 Dec;228:112112. doi: 10.1016/j.mad.2025.112112. Epub 2025 Sep 8.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

