 https://doi.org/10.4081/ejtm.2026.15026
https://doi.org/10.4081/ejtm.2026.15026
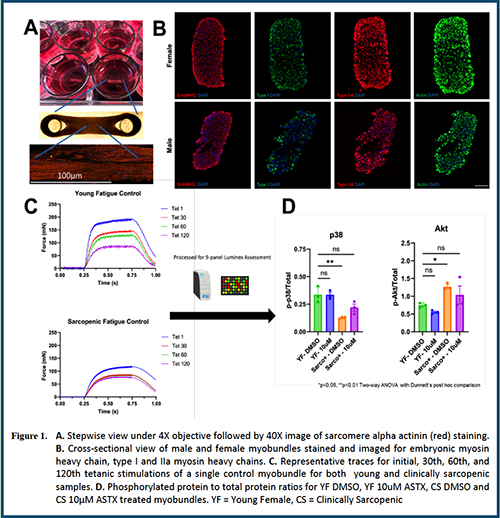
Abstract 027 | Investigating skeletal muscle diseases and therapeutics in a donor-derived bioengineered muscle platform
Karly A. Caples 1, Yangyi Edward Luo 2, Alexa Cooney 1, Elisabeth Barton 2, Karen Hecht 3, Siobhan Malany 1 | 1Department of Cellular and Systems Pharmacology, College of Pharmacy, University of Florida, Gainesville, FL, 32610; 2Department of Applied Physiology and Kinesiology, College of Health and Human Performance, University of Florida, Gainesville, FL, 32611 3AstraReal, Inc Karly A. 7761 Randolph Rd. NE, Moses Lake, WA 98837.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Börsch, A., Ham, D.J., Mittal, N. et al. Molecular and phenotypic analysis of rodent models reveals conserved and species-specific modulators of human sarcopenia. Commun Biol 4, 194 (2021). https://doi.org/10.1038/s42003-021-01723-z. DOI: https://doi.org/10.1038/s42003-021-01723-z
2. de Jong JCBC, Caspers MPM, Worms N, Keijzer N, Kleemann R, Menke AL, Nieuwenhuizen AG, Keijer J, Verschuren L, van den Hoek AM. Translatability of mouse muscle-aging for humans: the role of sex. Geroscience. 2024 Jun;46(3):3341-3360. doi: 10.1007/s11357-024-01082-7. Epub 2024 Jan 24. PMID: 38265577; PMCID: PMC11009184. DOI: https://doi.org/10.1007/s11357-024-01082-7
3. Lee SJ, Bhasin S, Klickstein L, Krishnan V, Rooks D. Challenges and Future Prospects of Targeting Myostatin/Activin A Signaling to Treat Diseases of Muscle Loss and Metabolic Dysfunction. J Gerontol A Biol Sci Med Sci. 2023 Jun 16;78(Suppl 1):32-37. doi: 10.1093/gerona/glad033. PMID: 36738276; PMCID: PMC10272974. DOI: https://doi.org/10.1093/gerona/glad033
4. Rybalka E, Timpani CA, Debruin DA, Bagaric RM, Campelj DG, Hayes A. The Failed Clinical Story of Myostatin Inhibitors against Duchenne Muscular Dystrophy: Exploring the Biology behind the Battle. Cells. 2020 Dec 10;9(12):2657. doi: 10.3390/cells9122657. PMID: 33322031; PMCID: PMC7764137. DOI: https://doi.org/10.3390/cells9122657
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

