 https://doi.org/10.4081/ejtm.2026.15024
https://doi.org/10.4081/ejtm.2026.15024
Abstract 025 | Proteomic profiling of the biofluid marker signature of dystrophinopathies
Kay Ohlendieck 1|2, Dieter Swandulla 3, Paul Dowling 1|2 | 1Department of Biology, Maynooth University, Maynooth, Co. Kildare, Ireland; 2Kathleen Lonsdale Institute for Human Health Research, Maynooth University, Maynooth, Co. Kildare, Ireland; 3Institute of Physiology, Faculty of Medicine, University of Bonn, Bonn, Germany.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Dowling P, Gargan S, Murphy S, Zweyer M, Sabir H, Swandulla D, Ohlendieck K. The Dystrophin Node as Integrator of Cytoskeletal Organization, Lateral Force Transmission, Fiber Stability and Cellular Signaling in Skeletal Muscle. Proteomes. 2021 Feb 2;9(1):9. doi: 10.3390/proteomes9010009. PMID: 33540575; PMCID: PMC7931087.
2. Dowling P, Swandulla D, Ohlendieck K. Cellular pathogenesis of Duchenne muscular dystrophy: progressive myofibre degeneration, chronic inflammation, reactive myofibrosis and satellite cell dysfunction. Eur J Transl Myol. 2023 Oct 16;33(4):11856. doi: 10.4081/ejtm.2023.11856. PMID: 37846661; PMCID: PMC10811648.
3. Zweyer M, Sabir H, Dowling P, Gargan S, Murphy S, Swandulla D, Ohlendieck K. Histopathology of Duchenne muscular dystrophy in correlation with changes in proteomic biomarkers. Histol Histopathol. 2022 Feb;37(2):101-116. doi: 10.14670/HH-18-403. Epub 2021 Dec 7. PMID: 34873679.
4. Dowling P, Swandulla D, Ohlendieck K. Mass Spectrometry-Based Proteomic Technology and Its Application to Study Skeletal Muscle Cell Biology. Cells. 2023 Nov 1;12(21):2560. doi: 10.3390/cells12212560. PMID: 37947638; PMCID: PMC10649384.
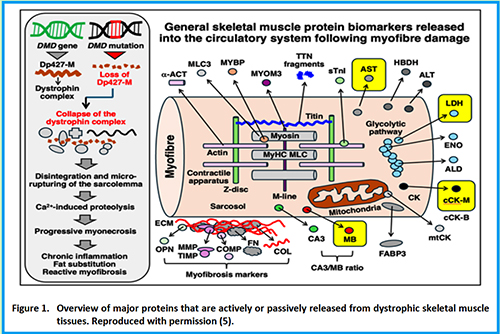
5. Dowling P, Negroni E, Trollet C, Zweyer M, Swandulla D, Ohlendieck K. Serum protein biomarker signature of Duchenne muscular dystrophy. Eur J Transl Myol. 2025 Jun 27;35(2):13956. doi: 10.4081/ejtm.2025.13956. Epub 2025 May 28. PMID: 40438995; PMCID: PMC12265423.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

