 https://doi.org/10.4081/ejtm.2026.15022
https://doi.org/10.4081/ejtm.2026.15022
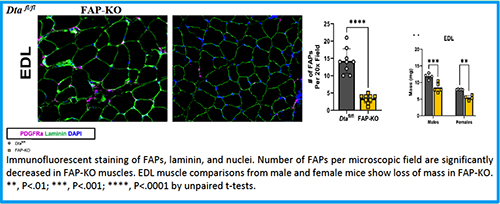
Abstract 023 | Fibro-adipogenic progenitors: gatekeepers for skeletal muscle mass maintenance
Yangyi E. Luo 1, Young il Lee2|3, Zoe Abe-Teh 1, Diana C. Muller 1, Lan Wei-LaPierre 1|3, Elisabeth R. Barton 1|3 | 1Department of Applied Physiology and Kinesiology, College of Health and Human Performance, University of Florida; 2Department of Pharmacology and Therapeutics, College of Medicine, University of Florida; 3Myology Institute, University of Florida, Gainesville, Florida, USA .
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Uezumi A, Fukada S, Yamamoto N, Takeda S, Tsuchida K. Mesenchymal progenitors distinct from satellite cells contribute to ectopic fat cell formation in skeletal muscle. Nat Cell Biol. 2010; 12(2): 143-52. PMID: 20081842.
2. Joe AW, Yi L, Natarajan A, Le Grand F, So L, Wang J, Rudnicki MA, Rossi FM. Muscle injury activates resident fibro/adipogenic progenitors that facilitate myogenesis. Nat Cell Biol. 2010;12(2): 153-63. PMID: 20081841. 2026 Pd3m Secretariate: Ugo Carraro – A-C M-C Foundation - Email: ugo.carraro@unipd.it - Ugo Carraro mobile: +39 338 1575745 – Barabara Ravara - A-C M-C Foundation - Email:barbara.ravara@unipd.it
3. Wosczyna MN, Konishi CT, Perez Carbajal EE, Wang TT, Walsh RA, Gan Q, Wagner MW, Rando TA. Mesenchymal Stromal Cells Are Required for Regeneration and Homeostatic Maintenance of Skeletal Muscle. Cell Rep. 2019; 27(7):2029-35 e5. PMID: 31091443.
4. Wu H, Mei L. Morphological analysis of neuromuscular junctions by immunofluorescent staining of whole-mount mouse diaphragms. Methods Mol Biol. 2013;1018: 277-85. PMID: 23681637.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

