 https://doi.org/10.4081/ejtm.2026.15021
https://doi.org/10.4081/ejtm.2026.15021
Abstract 022 | Lecture: new insights leading to improved designs of micro-dystrophins for use in adeno-associated viruses vectors
David Hammers, Cora Hart, Matthew Lee, H. Lee Sweeney, and UF Animal Assessment Core | University of Florida Myology Institute, Gainesville, Florida, USA.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
Downloads
1. Mendell, J. R. et al. Evidence-based path to newborn screening for Duchenne muscular dystrophy. Ann Neurol 71, 304-313, doi:10.1002/ana.23528 (2012). DOI: https://doi.org/10.1002/ana.23528
2. Hammers, D. W. et al. The D2.mdx mouse as a preclinical model of the skeletal muscle pathology associated with Duchenne muscular dystrophy. Sci Rep 10, 14070, doi:10.1038/s41598-020-70987-y (2020). DOI: https://doi.org/10.1038/s41598-020-70987-y
3. Fukada, S. et al. Genetic background affects properties of satellite cells and mdx phenotypes. Am J Pathol 176, 2414-2424, doi:10.2353/ajpath.2010.090887 (2010). DOI: https://doi.org/10.2353/ajpath.2010.090887
4. Coley, W. D. et al. Effect of genetic background on the dystrophic phenotype in mdx mice. Hum Mol Genet 25, 130-145, doi:10.1093/hmg/ddv460 (2016). DOI: https://doi.org/10.1093/hmg/ddv460
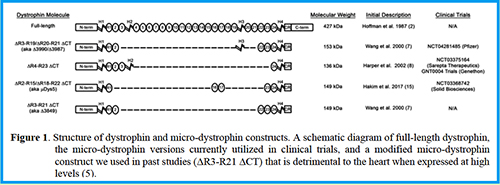
5. Hart CC, Lee YI, Xie J, Gao G, Lin BL, Hammers DW, Sweeney HL. (2024) Potential limitations of micro-dystrophin gene therapy for Duchenne muscular dystrophy. JCI Insight 9(11): e165869. DOI: https://doi.org/10.1172/jci.insight.165869
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

