 https://doi.org/10.4081/ejtm.2026.15008
https://doi.org/10.4081/ejtm.2026.15008
Abstract 009 | Lecture: FES in mice, rats and men: adaptation and recovery in aging rodent muscle
Jonathan Jarvis 1, Jack Edmondson 1, Hazel Sutherland 1, Mark Viggars 2 | 1School of Sport and Exercise Science, Liverpool John Moores University, UK; 2Department of Physiology & Aging – University of Florida, College of Medicine USA.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
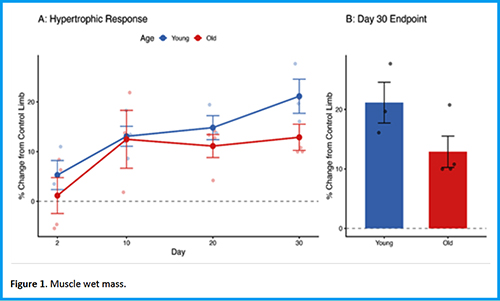
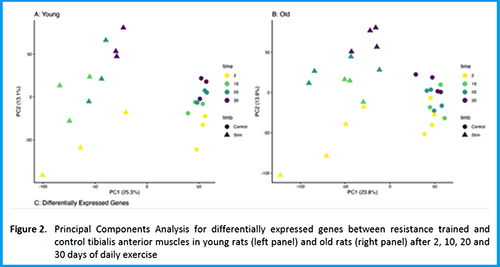
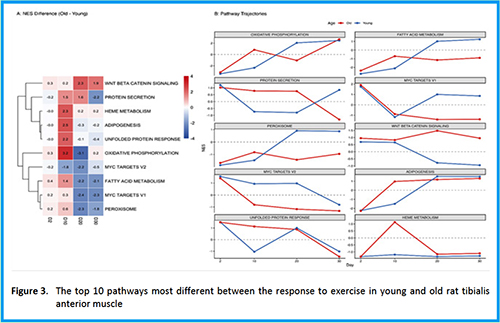
Downloads
1. Fisher AG, Seaborne RA, Hughes TM, Gutteridge A, Stewart C, Coulson JM. Sharples AP, Jarvis JC. Transcriptomic and epigenetic regulation of disuse atrophy and the return to activity in skeletal muscle. FASEB J. 2017 Dec;31(12):5268-5282. doi: 10.1096/fj.201700089RR. Epub 2017 Aug 17. PMID:28821632.
2. Viggars MR, Sutherland H, Lanmüller H, Schmoll M, Bijak M, Jarvis JC. Adaptation of the transcriptional response to resistance exercise over 4 weeks of daily training. FASEB J. 2023 Jan;37(1):e22686. doi: 10.1096/fj.202201418R. PMID: 36468768.
3. Stead CA, Hesketh SJ, Thomas ACQ, Viggars MR, Sutherland H, Jarvis JC, Burniston JG. Dynamic time course of muscle proteome adaptation to programmed resistance training in rats. Am J Physiol Cell Physiol. 2025 Dec 1;329(6):C1792-C1809. doi: 10.1152/ajpcell.00254.2025. Epub 2025 Oct 21. PMID: 41117415.
4. Viggars MR, Sutherland H, Cardozo CP, Jarvis JC. Conserved and species-specific transcriptional responses to daily programmed resistance exercise in rat and mouse. FASEB J. 2023 Dec;37(12):e23299. doi: 10.1096/fj.202301611R. PMID: 37994729.
5. Shavlakadze T, Xiong K, Mishra S, McEwen C, Gadi A, Wakai M, Salmon H, Stec MJ, Negron N, Ni M, Wei Y, Atwal GS, Bai Y, Glass DJ. Age-related gene expression signatures from limb skeletal muscles and the diaphragm in mice and rats reveal common and species-specific changes. Skelet Muscle. 2023 Jul 12;13(1):11. doi: 10.1186/s13395-023-00321-3. PMID: 37438807; PMCID: PMC10337157.
6. Brown A, Parise G, Thomas ACQ, Ng SY, McGlory C, Phillips SM, Kumbhare D, Joanisse S. Low baseline ribosome-related gene expression and resistance training-induced declines in ribosome-related gene expression are associated 2026 Pd3m Secretariate: Ugo Carraro – A-C M-C Foundation - Email: ugo.carraro@unipd.it - Ugo Carraro mobile: +39 338 1575745 – Barabara Ravara - A-C M-C Foundation - Email:barbara.ravara@unipd.it with skeletal muscle hypertrophy in young men and women. J Cell Physiol. 2024 Apr;239(4):e31182. doi: 10.1002/jcp.31182. Epub 2024 Jan 12. PMID: 38214457.
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

