 https://doi.org/10.4081/ejtm.2026.15000
https://doi.org/10.4081/ejtm.2026.15000
Abstract 001 | Lecture: human muscles are not just big mouse muscles
Richard L. Lieber1|2|3, Jan Fridén4 | 1Northwestern University, Chicago, IL, USA; 2Shirley Ryan AbilityLab, Chicago USA; 3Hines VA Medical Center, Chicago USA 4Swiss Paraplegic Center Nottwil, Switzerland.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 2 March 2026
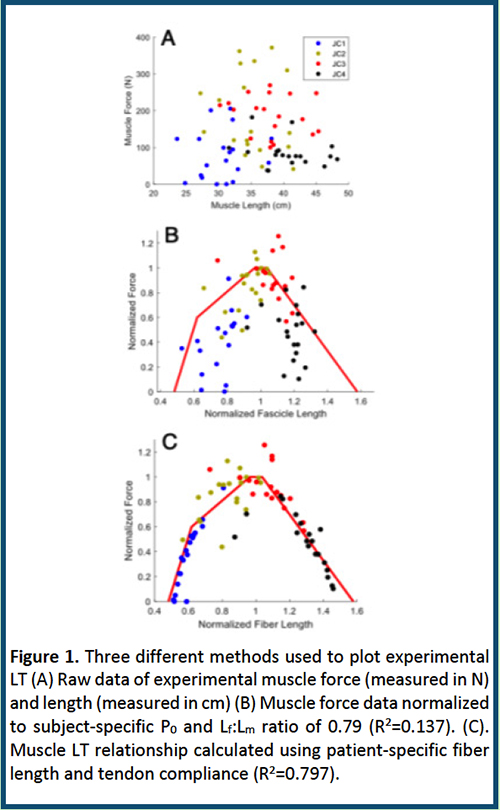
For over 30 years, we have been performing surgical research and reconstruction on human arms and legs for patients with spinal cord injuries (1), cerebral palsy (2), and overuse syndromes. Much of the basis for theories of how we design surgical procedures come from classic animal studies of the muscle sarcomere length-tension relationship for physiology, muscle architecture, for force and excursion predictions, and plasticity models to predict or explain adaptation to altered use. Unfortunately, predictions made based on these preclinical models have often proven false. During our studies, we have found many human muscle properties that do not simply “scale up” from mouse and other rodent muscles. Thus, we call for a new emphasis on the study of humans. While more complex, costly, and ethically challenging, we believe that this is a better use of resources to increase our world’s health. Human muscle scaling is unexpected. In the classic comparative physiology literature, it has clearly been demonstrated that muscle fiber length scales with body mass and bony dimensions (3). This comparative approach takes advantage of very large size differences amongst various species within the same family or very large size differences in animals with the same mode of locomotion, to reveal underlying design principles. Unfortunately, there has never been a large intraspecific study of human muscle architecture and thus, the way human muscle design varies with body size or the scaling laws that should be used in human musculoskeletal models are unknown. To address this issue, we created a large dataset of 896 muscles across 34 human lower extremities with a size range including approximately 90% and 70% of the United States population height and weight, respectively, across the range 36-103 years (4). The main result was clear—and confusing: While muscle mass scaled well with body mass, fiber length did not. Fiber length of individual muscles also did not correlate well with bone length A priori, we believed that it was reasonable to expect that muscle fiber length would scale with bone length or muscle insertion moment arm because joint moment arms, which determine the excursion of a muscle-tendon unit (MTU) scale with bone length and that moment arms geometrically scale with bone length (5). We have measured the relationship between human muscle sarcomere length and joint angle intraoperatively in human wrist flexors, extensors, quadriceps and lumbar multifidus muscles and have found that sarcomere length operating range is quite consistent across individuals. Given that muscle fibers represent sarcomeres arranged in series, similar sarcomere length operating range across individuals of varying size implies that serial sarcomere number (i.e., fiber length) would also scale with bony length or moment arm. This lack of scaling relationship has implications for biomechanical modeling studies in which architectural models are created based on a previously assumed linear relationship between fiber length and body segment length or body height. Human muscle length-tension properties are not well-predicted from architecture. Although we and others presented convincing data suggesting that skeletal muscle excursion is proportional to fascicle length and peak isometric force is proportional to physiological cross-sectional area this was not the case in our human experiments. We took advantage of our unique access to a large human muscle, the gracilis, which is transplanted into the arm to restore elbow flexion to patients with injury to the brachial plexus. After developing a buckle transducer method to measure isometric force (6), we measured the whole muscle length-tension curves in 19 patients. Interestingly, the raw length-tension relationshps were scattered with no resemblence to classic whole muscle length tension curves (Fig. 1A). While correction for muscle architecture and muscle length resulted in the rough expected shape of the curve, there was still a very large (86%) unexplained error (Fig. 1B). Since fiber length could not be predicted, we used the measured mechanical properties of each muscle and a predictor for patient-specific fiber length value. Using this value, ~80% of the experimental variability could be accounted for (Fig. 1C) which was completely consistent with the human sarcomere length-tension curve predicted by the sliding filament theory, correcting for human filament length. Human Sarcomere Length Adaptation is Inconsistent with Animal Models: It is well-established in animal models that serial sarcomere number increases when muscles are chronically stretched and decreases when muscles are chronically shortened. This principle has guided surgical reconstructive procedures for decades as surgeons try to restore function to “contractured” muscles or alter tendon transfer procedures based on the assumption that the muscle will “adjust” its length. Considering this dogma, imagine our surprise when we measured sarcomere length intraoperatively in patients with cerebral palsy and found that these chronically shortened muscles had extremely long sarcomeres (7, 8). This adaptation demonstrates that the factors that dictate muscle fiber length (serial sarcomere number) are dramatically disrupted with these types of injuries. The fact that dramatic structural adaptations are observed with this brain injury demonstrates a new dimension in understanding muscle adaptation—namely, that brain function and likely brain-induced growth factors, affect muscle properties in a way that is completely different than that observed in animal models. The mechanistic basis for this observation is only now being investigated (9, 10). Taken together, these studies reveal dramatic differences between animal models of muscle structure, function and plasticity and that observed in humans. We raise the call to increased study of human muscle and human disease to increase the health and fitness of our society.
Figure 1.
Downloads
1. Fridén J, Lieber RL. Reach out and grasp the opportunity: reconstructive hand surgery in tetraplegia. J Hand Surg Eur Vol. 2019;44(4):343-353. DOI: 10.1177/1753193419827814 DOI: https://doi.org/10.1177/1753193419827814
2. Lieber RL, Fridén J. Muscle contracture and passive mechanics in cerebral palsy. J Appl Physiol (1985). 2019;126(5):1492-1501. DOI: 10.1152/japplphysiol.00278.2018 DOI: https://doi.org/10.1152/japplphysiol.00278.2018
3. Alexander RM, Jayes AS, Maloiy GMO, Wathuta EM. Allometry of the leg muscles of mammals. J Zool Lond. 1981;194:539-552. DOI: https://doi.org/10.1111/j.1469-7998.1981.tb04600.x
4. Son J, Ward SR, Lieber RL. Scaling relationships between human leg muscle architectural properties and body size. J Exp Biol. 2024;227(6):jeb246567. DOI: 10.1242/jeb.246567 DOI: https://doi.org/10.1242/jeb.246567
5. Biewener AA. Allometry of quadrupedal locomotion: the scaling of duty factor, bone curvature and limb orientation to body size. J Exp Biol. 1983;105:147-171. DOI: https://doi.org/10.1242/jeb.105.1.147
6. Binder-Markey BI, Persad LS, Shin AY, Litchy WJ, Kaufman KR, Lieber RL. Direct intraoperative measurement of isometric contractile properties in living human muscle. J Physiol. 2023;601(10):1817-1830. DOI: 10.1113/JP284092 DOI: https://doi.org/10.1113/JP284092
7. Lieber RL, Fridén J. Spasticity causes a fundamental rearrangement of muscle-joint interaction. Muscle Nerve. 2002;25(2):265-270. DOI: 10.1002/mus.10036 DOI: https://doi.org/10.1002/mus.10036
8. Smith LR, Lee KS, Ward SR, Chambers HG, Lieber RL. Hamstring contractures in children with spastic cerebral palsy result from a stiffer extracellular matrix and increased in vivo sarcomere length. J Physiol. 2011;589(Pt 10):2625-2639. DOI: 10.1113/jphysiol.2010.203364 DOI: https://doi.org/10.1113/jphysiol.2010.203364
9. Domenighetti AA, Mathewson MA, Pichika R, Sibley LA, Zhao L, Chambers HG, Lieber RL. Loss of myogenic potential and fusion capacity of muscle stem cells isolated from contractured muscle in children with cerebral palsy. Am J Physiol Cell Physiol. 2018;315(2):C247-C257. DOI: 10.1152/ajpcell.00351.2017 DOI: https://doi.org/10.1152/ajpcell.00351.2017
10. Sibley LA, Broda N, Gross WR, Menezes AF, Embry RB, Swaroop VT, Chambers HG, Schipma MJ, Lieber RL, Domenighetti AA. Differential DNA methylation and transcriptional signatures characterize impairment of muscle stem cells in pediatric human muscle contractures after brain injury. FASEB J. 2021;35(10):e21928. DOI: 10.1096/fj.202100649R DOI: https://doi.org/10.1096/fj.202100649R
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

