 https://doi.org/10.4081/ejtm.2023.11555
https://doi.org/10.4081/ejtm.2023.11555
Proteomic profiling of the brain from the wobbler mouse model of amyotrophic lateral sclerosis reveals elevated levels of the astrogliosis marker glial fibrillary acidic protein
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 8 August 2023
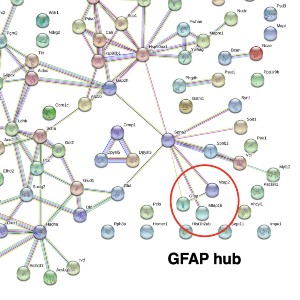
The wobbler mouse is a widely used model system of amyotrophic lateral sclerosis and exhibits progressive neurodegeneration and neuroinflammation in association with skeletal muscle wasting. This study has used wobbler brain preparations for the systematic and mass spectrometric determination of proteome-wide changes. The proteomic characterization of total protein extracts from wobbler specimens was carried out with the help of an Orbitrap mass spectrometer and revealed elevated levels of glia cell marker proteins, i.e., glial fibrillary acidic protein and the actin-binding protein coronin. In contrast, the abundance of the actin-binding protein neurabin and the scaffolding protein named piccolo of the presynaptic cytomatrix were shown to be reduced. The increased abundance of glial fibrillary acidic protein, which is frequently used in neuropathological studies as a marker protein of glial scar formation, was confirmed by immunoblotting. In analogy, the proteomic profiling of the brain from another established murine model of motor neuron disease, the SOD1mouse, also showed increased levels of this intermediate filament protein. This suggests that neurodegenerative processes are associated with astrogliosis in both the wobbler and SOD1 brain.
Downloads
Brown RH, Al-Chalabi A. Amyotrophic Lateral Sclerosis. N Engl J Med. 2017 Jul 13;377(2):162-172. PMID: 28700839. DOI: https://doi.org/10.1056/NEJMra1603471
Feldman EL, Goutman SA, Petri S, Mazzini L, Savelieff MG, Shaw PJ, Sobue G. Amyotrophic lateral sclerosis. Lancet. 2022 Oct 15;400(10360):1363-1380. Epub 2022 Sep 15. PMID: 36116464; PMCID: PMC10089700. DOI: https://doi.org/10.1016/S0140-6736(22)01272-7
Amado DA, Davidson BL. Gene therapy for ALS: A review. Mol Ther. 2021 Dec 1;29(12):3345-3358. Epub 2021 Apr 9. PMID: 33839324; PMCID: PMC8636154. DOI: https://doi.org/10.1016/j.ymthe.2021.04.008
Sever B, Ciftci H, DeMirci H, Sever H, Ocak F, Yulug B, Tateishi H, Tateishi T, Otsuka M, Fujita M, Başak AN. Comprehensive Research on Past and Future Therapeutic Strategies Devoted to Treatment of Amyotrophic Lateral Sclerosis. Int J Mol Sci. 2022 Feb 22;23(5):2400. PMID: 35269543; PMCID: PMC8910198. DOI: https://doi.org/10.3390/ijms23052400
Meijboom KE, Brown RH. Approaches to Gene Modulation Therapy for ALS. Neurotherapeutics. 2022 Jul;19(4):1159-1179. Epub 2022 Sep 6. PMID: 36068427; PMCID: PMC9587165. DOI: https://doi.org/10.1007/s13311-022-01285-w
Jaiswal MK. Riluzole and edaravone: A tale of two amyotrophic lateral sclerosis drugs. Med Res Rev. 2019 Mar;39(2):733-748. Epub 2018 Aug 12. PMID: 30101496. DOI: https://doi.org/10.1002/med.21528
Hardiman O, Al-Chalabi A, Chio A, Corr EM, Logroscino G, Robberecht W, Shaw PJ, Simmons Z, van den Berg LH. Amyotrophic lateral sclerosis. Nat Rev Dis Primers. 2017 Oct 5;3:17071. Erratum in: Nat Rev Dis Primers. 2017 Oct 20;3:17085. PMID: 28980624. DOI: https://doi.org/10.1038/nrdp.2017.85
Lui AJ, Byl NN. A systematic review of the effect of moderate intensity exercise on function and disease progression in amyotrophic lateral sclerosis. J Neurol Phys Ther. 2009 Jun;33(2):68-87. PMID: 19556916. DOI: https://doi.org/10.1097/NPT.0b013e31819912d0
Lunetta C, Lizio A, Sansone VA, Cellotto NM, Maestri E, Bettinelli M, Gatti V, Melazzini MG, Meola G, Corbo M. Strictly monitored exercise programs reduce motor deterioration in ALS: preliminary results of a randomized controlled trial. J Neurol. 2016 Jan;263(1):52-60. PMID: 26477027. DOI: https://doi.org/10.1007/s00415-015-7924-z
Clawson LL, Cudkowicz M, Krivickas L, Brooks BR, Sanjak M, Allred P, Atassi N, Swartz A, Steinhorn G, Uchil A, Riley KM, Yu H, Schoenfeld DA, Maragakis NJ; neals consortium. A randomized controlled trial of resistance and endurance exercise in amyotrophic lateral sclerosis. Amyotroph Lateral Scler Frontotemporal Degener. 2018 May;19(3-4):250-258. Epub 2017 Nov 30. PMID: 29191052. DOI: https://doi.org/10.1080/21678421.2017.1404108
Merico A, Cavinato M, Gregorio C, Lacatena A, Gioia E, Piccione F, Angelini C. ERRATUM: Effects of combined endurance and resistance training in Amyotrophic Lateral Sclerosis: A pilot, randomized, controlled study. Eur J Transl Myol. 2018 Sep 20;28(3):7842. Erratum for: Eur J Transl Myol. 2018 Mar 23;28(1):7278. PMID: 30344983; PMCID: PMC6176392. DOI: https://doi.org/10.4081/ejtm.2018.7842
Chaudhary R, Agarwal V, Rehman M, Kaushik AS, Mishra V. Genetic architecture of motor neuron diseases. J Neurol Sci. 2022 Mar 15;434:120099. Epub 2021 Dec 22. PMID: 34965490. DOI: https://doi.org/10.1016/j.jns.2021.120099
Akçimen F, Lopez ER, Landers JE, Nath A, Chiò A, Chia R, Traynor BJ. Amyotrophic lateral sclerosis: translating genetic discoveries into therapies. Nat Rev Genet. 2023 Apr 6. Epub ahead of print. PMID: 37024676. DOI: https://doi.org/10.1038/s41576-023-00592-y
Witzel S, Mayer K, Oeckl P. Biomarkers for amyotrophic lateral sclerosis. Curr Opin Neurol. 2022 Oct 1;35(5):699-704. Epub 2022 Aug 4. PMID: 35942674. DOI: https://doi.org/10.1097/WCO.0000000000001094
Schmitt-John T. VPS54 and the wobbler mouse. Front Neurosci. 2015 Oct 21;9:381. PMID: 26539077; PMCID: PMC4612502. DOI: https://doi.org/10.3389/fnins.2015.00381
Schmitt-John T, Drepper C, Mussmann A, Hahn P, Kuhlmann M, Thiel C, Hafner M, Lengeling A, Heimann P, Jones JM, Meisler MH, Jockusch H. Mutation of Vps54 causes motor neuron disease and defective spermiogenesis in the wobbler mouse. Nat Genet. 2005 Nov;37(11):1213-5. Epub 2005 Oct 23. PMID: 16244655. DOI: https://doi.org/10.1038/ng1661
Moser JM, Bigini P, Schmitt-John T. The wobbler mouse, an ALS animal model. Mol Genet Genomics. 2013 Jun;288(5-6):207-29. Epub 2013 Mar 29. PMID: 23539154; PMCID: PMC3664746. DOI: https://doi.org/10.1007/s00438-013-0741-0
Pérez-Victoria FJ, Abascal-Palacios G, Tascón I, Kajava A, Magadán JG, Pioro EP, Bonifacino JS, Hierro A. Structural basis for the wobbler mouse neurodegenerative disorder caused by mutation in the Vps54 subunit of the GARP complex. Proc Natl Acad Sci U S A. 2010 Jul 20;107(29):12860-5. Epub 2010 Jul 6. PMID: 20615984; PMCID: PMC2919957. DOI: https://doi.org/10.1073/pnas.1004756107
Meyer M, Lima A, Deniselle MCG, De Nicola AF. Early Signs of Neuroinflammation in the Postnatal Wobbler Mouse Model of Amyotrophic Lateral Sclerosis. Cell Mol Neurobiol. 2023 Jul;43(5):2149-2163. Epub 2022 Oct 11. PMID: 36219378. DOI: https://doi.org/10.1007/s10571-022-01294-5
Staunton L, Jockusch H, Ohlendieck K. Proteomic analysis of muscle affected by motor neuron degeneration: the wobbler mouse model of amyotrophic lateral sclerosis. Biochem Biophys Res Commun. 2011 Mar 25;406(4):595-600. Epub 2011 Feb 24. PMID: 21354103. DOI: https://doi.org/10.1016/j.bbrc.2011.02.099
Jockusch H, Holland A, Staunton L, Schmitt-John T, Heimann P, Dowling P, Ohlendieck K. Pathoproteomics of testicular tissue deficient in the GARP component VPS54: the wobbler mouse model of globozoospermia. Proteomics. 2014 Apr;14(7-8):839-52. Epub 2013 Nov 13. PMID: 24115398. DOI: https://doi.org/10.1002/pmic.201300189
Holland A, Schmitt-John T, Dowling P, Meleady P, Henry M, Clynes M, Ohlendieck K. Intricate effects of primary motor neuronopathy on contractile proteins and metabolic muscle enzymes as revealed by label-free mass spectrometry. Biosci Rep. 2014 Jul 1;34(4):e00119. PMID: 24895011; PMCID: PMC4076836. DOI: https://doi.org/10.1042/BSR20140029
Capitanio D, Vasso M, Ratti A, Grignaschi G, Volta M, Moriggi M, Daleno C, Bendotti C, Silani V, Gelfi C. Molecular signatures of amyotrophic lateral sclerosis disease progression in hind and forelimb muscles of an SOD1(G93A) mouse model. Antioxid Redox Signal. 2012 Nov 15;17(10):1333-50. Epub 2012 Jun 13. PMID: 22563797; PMCID: PMC3437050. DOI: https://doi.org/10.1089/ars.2012.4524
Murphy S, Zweyer M, Henry M, Meleady P, Mundegar RR, Swandulla D, Ohlendieck K. Label-free mass spectrometric analysis reveals complex changes in the brain proteome from the mdx-4cv mouse model of Duchenne muscular dystrophy. Clin Proteomics. 2015 Nov 23;12:27. PMID: 26604869; PMCID: PMC4657206. DOI: https://doi.org/10.1186/s12014-015-9099-0
Dowling P, Gargan S, Zweyer M, Henry M, Meleady P, Swandulla D, Ohlendieck K. Protocol for the Bottom-Up Proteomic Analysis of Mouse Spleen. STAR Protoc. 2020 Dec 3;1(3):100196. PMID: 33377090; PMCID: PMC7757555. DOI: https://doi.org/10.1016/j.xpro.2020.100196
Dowling P, Gargan S, Zweyer M, Sabir H, Henry M, Meleady P, Swandulla D, Ohlendieck K. Proteomic profiling of the interface between the stomach wall and the pancreas in dystrophinopathy. Eur J Transl Myol. 2021 Mar 26;31(1):9627. PMID: 33709651; PMCID: PMC8056161. DOI: https://doi.org/10.4081/ejtm.2021.9627
Dowling P, Gargan S, Zweyer M, Henry M, Meleady P, Swandulla D, Ohlendieck K. Proteome-wide Changes in the mdx-4cv Spleen due to Pathophysiological Cross Talk with Dystrophin-Deficient Skeletal Muscle. iScience. 2020 Aug 26;23(9):101500. Epub ahead of print. PMID: 32916630; PMCID: PMC7490529. DOI: https://doi.org/10.1016/j.isci.2020.101500
Szklarczyk D, Kirsch R, Koutrouli M, Nastou K, Mehryary F, Hachilif R, Gable AL, Fang T, Doncheva NT, Pyysalo S, Bork P, Jensen LJ, von Mering C. The STRING database in 2023: protein-protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 2023 Jan 6;51(D1):D638-D646. PMID: 36370105; PMCID: PMC9825434. DOI: https://doi.org/10.1093/nar/gkac1000
Taraslia VK, Kouskoukis A, Anagnostopoulos AK, Stravopodis DJ, Margaritis LH, Tsangaris GT. Proteomic analysis of normal murine brain parts. Cancer Genomics Proteomics. 2013 May-Jun;10(3):125-54. PMID: 23741028.
Sharma K, Schmitt S, Bergner CG, Tyanova S, Kannaiyan N, Manrique-Hoyos N, Kongi K, Cantuti L, Hanisch UK, Philips MA, Rossner MJ, Mann M, Simons M. Cell type- and brain region-resolved mouse brain proteome. Nat Neurosci. 2015 Dec;18(12):1819-31. Epub 2015 Nov 2. PMID: 26523646; PMCID: PMC7116867. DOI: https://doi.org/10.1038/nn.4160
Davis RG, Park HM, Kim K, Greer JB, Fellers RT, LeDuc RD, Romanova EV, Rubakhin SS, Zombeck JA, Wu C, Yau PM, Gao P, van Nispen AJ, Patrie SM, Thomas PM, Sweedler JV, Rhodes JS, Kelleher NL. Top-Down Proteomics Enables Comparative Analysis of Brain Proteoforms Between Mouse Strains. Anal Chem. 2018 Mar 20;90(6):3802-3810. 7b04108. Epub 2018 Feb 26. PMID: 29481055; PMCID: PMC5861018. DOI: https://doi.org/10.1021/acs.analchem.7b04108
Ghanavatinejad F, Fard Tabrizi ZP, Omidghaemi S, Sharifi E, Møller SG, Jami MS. Protein biomarkers of neural system. J Otol. 2019 Sep;14(3):77-88. Epub 2019 Mar 23. PMID: 31467504; PMCID: PMC6712353. DOI: https://doi.org/10.1016/j.joto.2019.03.001
Korovesi AG, Anagnostopoulos AK, Pierros V, Stravopodis DJ, Tsangaris GT. Normal Mouse Brain Proteome II: Analysis of Brain Regions by High-resolution Mass Spectrometry. Cancer Genomics Proteomics. 2020 Nov-Dec;17(6):757-767. PMID: 33099477; PMCID: PMC7675658. DOI: https://doi.org/10.21873/cgp.20230
Dowling P, Zweyer M, Sabir H, Henry M, Meleady P, Swandulla D, Ohlendieck K. Mass spectrometry-based proteomic characterization of the middle-aged mouse brain for animal model research of neuromuscular diseases. Eur J Transl Myol. 2023;11553. DOI: https://doi.org/10.4081/ejtm.2023.11553
Yang Z, Wang KK. Glial fibrillary acidic protein: from intermediate filament assembly and gliosis to neurobiomarker. Trends Neurosci. 2015 Jun;38(6):364-74. Epub 2015 May 11. PMID: 25975510; PMCID: PMC4559283. DOI: https://doi.org/10.1016/j.tins.2015.04.003
Ahmed Z, Shaw G, Sharma VP, Yang C, McGowan E, Dickson DW. Actin-binding proteins coronin-1a and IBA-1 are effective microglial markers for immunohistochemistry. J Histochem Cytochem. 2007 Jul;55(7):687-700. Epub 2007 Mar 6. PMID: 17341475. DOI: https://doi.org/10.1369/jhc.6A7156.2007
Karlsson P, Droce A, Moser JM, Cuhlmann S, Padilla CO, Heimann P, Bartsch JW, Füchtbauer A, Füchtbauer EM, Schmitt-John T. Loss of vps54 function leads to vesicle traffic impairment, protein mis-sorting and embryonic lethality. Int J Mol Sci. 2013 May 24;14(6):10908-25. PMID: 23708095; PMCID: PMC3709709. DOI: https://doi.org/10.3390/ijms140610908
Thielsen KD, Moser JM, Schmitt-John T, Jensen MS, Jensen K, Holm MM. The Wobbler mouse model of amyotrophic lateral sclerosis (ALS) displays hippocampal hyperexcitability, and reduced number of interneurons, but no presynaptic vesicle release impairments. PLoS One. 2013 Dec 11;8(12):e82767. Erratum in: PLoS One. 2015;10(3):e0121772. PMID: 24349357; PMCID: PMC3859636. DOI: https://doi.org/10.1371/journal.pone.0082767
Saberi D, Ott B, Dahlke C, Matschke V, Schmitt-John T, Theiss C. The Spatiotemporal Pattern of Degeneration in the Cerebellum of the Wobbler Mouse. J Neuropathol Exp Neurol. 2016 Apr;75(4):347-57. Epub 2016 Mar 4. PMID: 26945034. DOI: https://doi.org/10.1093/jnen/nlw005
Andreasen SR, Lundbye CJ, Christensen TB, Thielsen KD, Schmitt-John T, Holm MM. Excitatory-inhibitory imbalance in the brain of the wobbler mouse model of amyotrophic lateral sclerosis substantiated by riluzole and diazepam. Neurosci Lett. 2017 Sep 29;658:85-90. Epub 2017 Aug 18. PMID: 28823891. DOI: https://doi.org/10.1016/j.neulet.2017.08.033
Hol EM, Pekny M. Glial fibrillary acidic protein (GFAP) and the astrocyte intermediate filament system in diseases of the central nervous system. Curr Opin Cell Biol. 2015 Feb;32:121-30. Epub 2015 Mar 2. PMID: 25726916. DOI: https://doi.org/10.1016/j.ceb.2015.02.004
Mustafov D, Karteris E, Braoudaki M. Deciphering the Role of microRNA Mediated Regulation of Coronin 1C in Glioblastoma Development and Metastasis. Noncoding RNA. 2023 Jan 4;9(1):4.. PMID: 36649032; PMCID: PMC9844418. DOI: https://doi.org/10.3390/ncrna9010004
Qian K, Jiang X, Liu ZQ, Zhang J, Fu P, Su Y, Brazhe NA, Liu D, Zhu LQ. Revisiting the critical roles of reactive astrocytes in neurodegeneration. Mol Psychiatry. 2023 Apr 10. Epub ahead of print. PMID: 37037874. DOI: https://doi.org/10.1038/s41380-023-02061-8
Purushotham SS, Buskila Y. Astrocytic modulation of neuronal signalling. Front Netw Physiol. 2023 Jun 1;3:1205544. PMID: 37332623; PMCID: PMC10269688. DOI: https://doi.org/10.3389/fnetp.2023.1205544
Ghasemi M, Keyhanian K, Douthwright C. Glial Cell Dysfunction in C9orf72-Related Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Cells. 2021 Jan 28;10(2):249. PMID: 33525344; PMCID: PMC7912327. DOI: https://doi.org/10.3390/cells10020249
Guttenplan KA, Weigel MK, Adler DI, Couthouis J, Liddelow SA, Gitler AD, Barres BA. Knockout of reactive astrocyte activating factors slows disease progression in an ALS mouse model. Nat Commun. 2020 Jul 27;11(1):3753. PMID: 32719333; PMCID: PMC7385161. DOI: https://doi.org/10.1038/s41467-020-17514-9
Corcia P, Lunetta C, Vourc'h P, Pradat PF, Blasco H. Time for optimism in amyotrophic lateral sclerosis. Eur J Neurol. 2023 May;30(5):1459-1464. Epub 2023 Feb 23. PMID: 36773012. DOI: https://doi.org/10.1111/ene.15738
Vu LT, Bowser R. Fluid-Based Biomarkers for Amyotrophic Lateral Sclerosis. Neurotherapeutics. 2017 Jan;14(1):119-134. PMID: 27933485; PMCID: PMC5233638. DOI: https://doi.org/10.1007/s13311-016-0503-x
Verde F, Otto M, Silani V. Neurofilament Light Chain as Biomarker for Amyotrophic Lateral Sclerosis and Frontotemporal Dementia. Front Neurosci. 2021 Jun 21;15:679199. PMID: 34234641; PMCID: PMC8255624. DOI: https://doi.org/10.3389/fnins.2021.679199
Abu-Rumeileh S, Vacchiano V, Zenesini C, Polischi B, de Pasqua S, Fileccia E, Mammana A, Di Stasi V, Capellari S, Salvi F, Liguori R, Parchi P; BoReALS. Diagnostic-prognostic value and electrophysiological correlates of CSF biomarkers of neurodegeneration and neuroinflammation in amyotrophic lateral sclerosis. J Neurol. 2020 Jun;267(6):1699-1708. Epub 2020 Feb 25. PMID: 32100123. DOI: https://doi.org/10.1007/s00415-020-09761-z
Gaiani A, Martinelli I, Bello L, Querin G, Puthenparampil M, Ruggero S, Toffanin E, Cagnin A, Briani C, Pegoraro E, Sorarù G. Diagnostic and Prognostic Biomarkers in Amyotrophic Lateral Sclerosis: Neurofilament Light Chain Levels in Definite Subtypes of Disease. JAMA Neurol. 2017 May 1;74(5):525-532. PMID: 28264096; PMCID: PMC5822207. DOI: https://doi.org/10.1001/jamaneurol.2016.5398
Verde F, Milone I, Colombo E, Maranzano A, Solca F, Torre S, Doretti A, Gentile F, Manini A, Bonetti R, Peverelli S, Messina S, Maderna L, Morelli C, Poletti B, Ratti A, Silani V, Ticozzi N. Phenotypic correlates of serum neurofilament light chain levels in amyotrophic lateral sclerosis. Front Aging Neurosci. 2023 Mar 15;15:1132808. PMID: 37009451; PMCID: PMC10050442. DOI: https://doi.org/10.3389/fnagi.2023.1132808
Verde F, Milone I, Maranzano A, Colombo E, Torre S, Solca F, Doretti A, Gentile F, Manini A, Bonetti R, Peverelli S, Messina S, Maderna L, Morelli C, Poletti B, Ratti A, Silani V, Ticozzi N. Serum levels of glial fibrillary acidic protein in patients with amyotrophic lateral sclerosis. Ann Clin Transl Neurol. 2023 Jan;10(1):118-129. Epub 2022 Dec 16. PMID: 36525477; PMCID: PMC9852391. DOI: https://doi.org/10.1002/acn3.51708
Falzone YM, Domi T, Mandelli A, Pozzi L, Schito P, Russo T, Barbieri A, Fazio R, Volontè MA, Magnani G, Del Carro U, Carrera P, Malaspina A, Agosta F, Quattrini A, Furlan R, Filippi M, Riva N. Integrated evaluation of a panel of neurochemical biomarkers to optimize diagnosis and prognosis in amyotrophic lateral sclerosis. Eur J Neurol. 2022 Jul;29(7):1930-1939. Epub 2022 Mar 23. PMID: 35263489; PMCID: PMC9314044. DOI: https://doi.org/10.1111/ene.15321
Bede P, Lope J. Biomarker development in amyotrophic lateral sclerosis: Challenges and viable strategies. Eur J Neurol. 2022 Jul;29(7):1867-1868. Epub 2022 May 4. PMID: 35467797. DOI: https://doi.org/10.1111/ene.15372
Roeben B, Wilke C, Bender B, Ziemann U, Synofzik M. The motor band sign in ALS: presentations and frequencies in a consecutive series of ALS patients. J Neurol Sci. 2019 Nov 15;406:116440. Epub 2019 Aug 30. PMID: 31521959. DOI: https://doi.org/10.1016/j.jns.2019.116440
Valaparla VL, Lobaina M, Patel C, Patel AV. Motor Band Sign in Primary Lateral Sclerosis: A Case Report Proposing the Need for an Imaging Biomarker. Cureus. 2023 Mar 14;15(3):e36121. PMID: 37065386; PMCID: PMC10101188. DOI: https://doi.org/10.7759/cureus.36121
Chung HS, Melkus G, Bourque P, Chakraborty S. Motor Band Sign in Motor Neuron Disease: A Marker for Upper Motor Neuron Involvement. Can J Neurol Sci. 2023 May;50(3):373-379. Epub 2022 Apr 28. PMID: 35477836. DOI: https://doi.org/10.1017/cjn.2022.52
How to Cite

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.

