A serious fit of cough: A 66-year-old patient with myasthenia gravis presenting with spontaneous intercostal lung herniation after coughing. Case report and pathophysiological discussion
Accepted: 18 May 2020
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
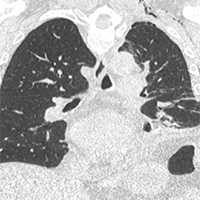
We present a case of a 66-year-old man with history of myasthenia gravis, severe obesity and osteopenia self-presented to our Emergency Department (ED) with severe pain in his left hemithorax, occurred after an episode of cough three days before. No history of trauma was reported. The patient underwent a chest-XR showing uncomplicated spontaneous fractures of the 5th and 6th left ribs. He was therefore discharged with appropriate analgesic treatment. Five days later, the patient came back to our ED for a wide left abdominal hematoma, though hemodynamically stable and eupneic. A CT-scan with contrast showed a rare and unexpected spontaneous left intercostal lung herniation complicated with a diffuse subcutaneous emphysema, pneumothorax, loculated bilateral pleural effusion and abdominal hematoma. The patient was admitted, treated conservatively and safely discharged after two weeks. We also provide a pathophysiological discussion of the case and a literature review.
M. K. Karmakar and A. M.-H. Ho, “Acute pain management of patients with multiple fractured ribs.,” J. Trauma, vol. 54, no. 3, pp. 615–25, Mar. 2003. DOI: https://doi.org/10.1097/01.TA.0000053197.40145.62
B. S. Talbot, C. P. Gange Jr., A. Chaturvedi, N. Klionsky, S. K. Hobbs, and A. Chaturvedi, “Traumatic Rib Injury: Patterns, Imaging Pitfalls, Complications, and Treatment.[Erratum appears in Radiographics. 2017 May-Jun;37(3):1004; PMID: 28493796],” Radiographics, vol. 37, no. 2, pp. 628–651, 2017.
T. Maeda, R. Sato, S. K. Luthe, and M. C. Russell, “Spontaneous Intercostal Lung Hernia.,” Am. J. Med., vol. 130, no. 9, pp. e399–e400, Sep. 2017. DOI: https://doi.org/10.1016/j.amjmed.2017.03.053
A. M. O’ Mahony, K. M. Murphy, T. M. O’Connor, and D. R. Curran, “Spontaneous pulmonary hernia secondary to intercostal muscle tear,” BMJ Case Rep., vol. 12, no. 10, Oct. 2019. DOI: https://doi.org/10.1136/bcr-2019-231706
F. C. Y. Lee, “The Curtain Sign in Lung Ultrasound,” J. Med. Ultrasound, vol. 25, no. 2, pp. 101–104, Jun. 2017. DOI: https://doi.org/10.1016/j.jmu.2017.04.005
R. J. Hallifax, A. Talwar, J. M. Wrightson, A. Edey, and F. V. Gleeson, “State-of-the-art: Radiological investigation of pleural disease,” Respiratory Medicine, vol. 124. W.B. Saunders Ltd, pp. 88–99, 01-Mar-2017. DOI: https://doi.org/10.1016/j.rmed.2017.02.013
M. Cox, D. Thota, and R. Trevino, “Spontaneous Lung Herniation Through the Chest Wall,” Mil. Med., vol. 183, no. 3–4, pp. e233–e234, Mar. 2018. DOI: https://doi.org/10.1093/milmed/usx063
G. S. Gaude, R. Chatterji, A. S. Bagga, A. G. Naik, and V. Bellad, “Spontaneous lung hernia.,” Indian J. Chest Dis. Allied Sci., vol. 39, no. 1, pp. 65–68, 1997.
R. T. Ross and C. M. Burnett, “Atraumatic lung hernia.,” Ann. Thorac. Surg., vol. 67, no. 5, pp. 1496–7, May 1999.
H. Haneda, K. Okuda, and R. Nakanishi, “Case of intercostal lung hernia with hemosputum that developed after thoracoscopic lobectomy,” Asian J. Endosc. Surg., vol. 12, no. 4, pp. 449–451, Oct. 2019. DOI: https://doi.org/10.1111/ases.12656
A. Haro et al., “A rare case of an intercostal lung herniation with fractures of the fifth and sixth ribs after thoracic surgery,” Int. J. Surg. Case Rep., vol. 41, pp. 473–476, Jan. 2017. DOI: https://doi.org/10.1016/j.ijscr.2017.11.028
L. C. Hofbauer et al., “Stimulation of Osteoprotegerin Ligand and Inhibition of Osteoprotegerin Production by Glucocorticoids in Human Osteoblastic Lineage Cells: Potential Paracrine Mechanisms of Glucocorticoid-Induced Osteoporosis 1,” Endocrinology, vol. 140, no. 10, pp. 4382–4389, Oct. 1999.
S. Khosla, “Minireview: The OPG/RANKL/RANK System,” Endocrinology, vol. 142, no. 12, pp. 5050–5055, Dec. 2001.
R. S. Weinstein, R. L. Jilka, A. Michael Parfitt, and S. C. Manolagas, “Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts end osteocytes by glucocorticoids potential mechanisms of their deleterious effects on bone,” J. Clin. Invest., vol. 102, no. 2, pp. 274–282, Jul. 1998. DOI: https://doi.org/10.1172/JCI2799
R. S. Weinstein, “Clinical practice. Glucocorticoid-induced bone disease,” N. Engl. J. Med., vol. 365, no. 1, pp. 62–70, Jul. 2011. DOI: https://doi.org/10.1056/NEJMcp1012926
T. Yang, Z. Li, L. Jiang, and X. Xi, “Corticosteroid use and intensive care unit-acquired weakness: a systematic review and meta-analysis,” Crit. Care, vol. 22, no. 1, p. 187, Dec. 2018. DOI: https://doi.org/10.1186/s13054-018-2111-0
Y. Gupta and A. Gupta, “Glucocorticoid-induced myopathy: Pathophysiology, diagnosis, and treatment,” Indian J. Endocrinol. Metab., vol. 17, no. 5, p. 913, 2013. DOI: https://doi.org/10.4103/2230-8210.117215
N. Behazin, S. B. Jones, R. I. Cohen, and S. H. Loring, “Respiratory restriction and elevated pleural and esophageal pressures in morbid obesity,” J. Appl. Physiol., vol. 108, no. 1, pp. 212–218, Jan. 2010. DOI: https://doi.org/10.1152/japplphysiol.91356.2008
N. E. Gilhus and J. J. Verschuuren, “Myasthenia gravis: Subgroup classification and therapeutic strategies,” The Lancet Neurology, vol. 14, no. 10. Lancet Publishing Group, pp. 1023–1036, 01-Oct-2015.
D. Drachman, “Myasthenia Gravis,” Semin. Neurol., vol. 36, no. 05, pp. 419–424, Sep. 2016. DOI: https://doi.org/10.1055/s-0036-1586265
B. Berry et al., “Acquired intercostal lung herniation: conservative management may lead to continuation of symptoms and other adverse consequence,” Gen. Thorac. Cardiovasc. Surg., pp. 1–5, Jun. 2019. DOI: https://doi.org/10.1007/s11748-019-01156-w
G. Batıhan, D. Yaldız, and K. C. Ceylan, “A rare complication of video-assisted thoracoscopic surgery: lung herniation retrospective case series of three patients and review of the literature.,” Wideochirurgia i inne Tech. maloinwazyjne = Videosurgery other miniinvasive Tech., vol. 15, no. 1, pp. 215–219, Mar. 2020. DOI: https://doi.org/10.5114/wiitm.2019.87937
B. François, A. Desachy, E. Cornu, E. Ostyn, L. Niquet, and P. Vignon, “Traumatic pulmonary hernia: surgical versus conservative management.,” J. Trauma, vol. 44, no. 1, pp. 217–9, Jan. 1998. DOI: https://doi.org/10.1097/00005373-199801000-00035
D. Wussler et al., “Clinical Utility of Procalcitonin in the Diagnosis of Pneumonia.,” Clin. Chem., Oct. 2019.
A. Çolak, C. Yllmaz, B. Toprak, and S. Aktoǧu, “Procalcitonin and CRP as Biomarkers in Discrimination of Community-Acquired Pneumonia and Exacerbation of COPD,” J. Med. Biochem., vol. 36, no. 2, pp. 122–126, Apr. 2017. DOI: https://doi.org/10.1515/jomb-2017-0011
PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.


 https://doi.org/10.4081/ecj.2020.8911
https://doi.org/10.4081/ecj.2020.8911







