Modulation of hydrolytic profiles of cell-bound and cell-free exoenzymes in Antarctic marine bacterial isolates
Cell-bound and cell-free enzymatic profiles of pelagic marine bacteria

Accepted: 18 June 2019
HTML: 31
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
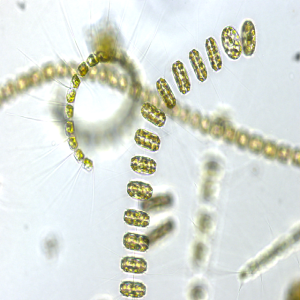
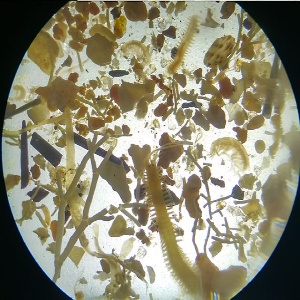
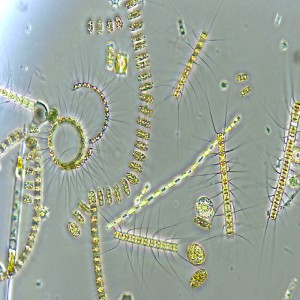
Microbial degradation of high molecular weight organic matter in the marine environment is dependent on the synthesis and activity of hydrolytic exoenzymes. These can be found both associated to the cell (i.e., attached to cell-wall or in the periplasmic space) and cell-free, dissolved in the surrounding waters. Recent evidences suggest that exoenzymes location is linked to different foraging strategies. ‘Selfish’ bacteria use cell-bound enzymes to obtain large oligomers which are then further degraded in the confined periplasmic space, whereas cell-free enzymes are thought to be advantageous for particle-attached prokaryotes, consequently ‘sharing’ the hydrolysis product with other members of the degrading consortium. We examined whether different bacterial isolates exhibit different exoenzymatic activity profiles when exposed to different growth condition. Seven bacterial strains, isolated from the Ross Sea, were screened for the production of β-glucosidase, alkaline-phosphatase, lipase, chitinase and leucine aminopeptidase, assaying the hydrolysis rates of both cell-bound and cell-free exoenzymatic fractions. Furthermore, to test whether bacteria growing on particles are more prone to produce cell-free exoenzymes, we set up a controlled experiment, amending one of the bacterial isolates with phytodetritus, in order to promote the colonization of aggregates. Cell specific hydrolytic rates were highly variable and different isolates expressed different dominant hydrolytic activities. These results demonstrate a specialization for different substrates in different bacteria, further suggesting that some of the tested isolates may have an intrinsic potential to copiously produce cell-free exoenzymes. Moreover, our results suggest that this specialization is not strictly genetically determined but varies according to growth conditions. The experiment performed with phytodetrital particles highlighted an increasing contribution of the dissolved exoenzymatic activity in samples bearing aggregates and associated bacteria. Also, the tested isolate further modulated its hydrolytic machinery, expressing a different enzymatic profile when exposed to phytodetrital particles. The continuation of these experimental activities, testing the response of different bacterial isolates to different substrates, will help to determine the conditions under which different hydrolysis patterns develop, deepening the current knowledge on the organic matter cycling in the ocean.
Supporting Agencies
Italian Ministry for Education University and Research MIUR, Italian Program for Research in Antarctica (PNRA) with funds from the project PRIAMO (PRokaryotes Interactions with Antarctic phytodetritus: a Micro- to macroscale voyage from the surface to the deep Ocean), PNRA16_00103PAGEPress has chosen to apply the Creative Commons Attribution NonCommercial 4.0 International License (CC BY-NC 4.0) to all manuscripts to be published.



 https://doi.org/10.4081/aiol.2019.8240
https://doi.org/10.4081/aiol.2019.8240